In the last couple of years, in an effort of Making TPN compounding safe, we have recorded a serious upswing and increase in the use of automated IV preparation systems. Their popularity is directly linked to their ease of use since they combine barcode scanning and Digital image capture to ensure safer and more efficient compounding processes. With the increase of use of such technology, we’ve seen an increase in the risks and sources of contamination of compounded products when prepared with an IV automation workflow machine. Currently, there are many TPN compounder manufacturers on the market, such as Epic, Baxter, Baxa, MedKeeper, Omnicell, ICU medical, and many other players in this segment.
We’ve been carefully monitoring the responses regarding these types of machinery whenever there was an inspection by the FDA at a facility that houses and uses such technology. Pretty much all oversights noticed issues with the proper placement of the IV compounder in the working area; issues that either exposed the operator to risks or cases where the technician contaminated the product.
With this information in hand, we decided to review the IV TPN preparation procedure, SOP, and workflow setups. So please read carefully the following article and let us know what you think by leaving a comment in the comment section at the very end of the page.
Every time a needle pierces a vial’s rubber top or the injection port of an IV bag, there is a risk of contamination of the CSP, because TPN solutions are comprised of multiple solution components mixed with multiple additive components, the risk of contamination is greater with TPNs than it is with other CSPs. This risk is well addressed in the revision of the USP 797 where the Sterile compounding and proper aseptic techniques are explained for easier application for Pharmacy Technicians.
When called upon a project after a site inspection we discovered multiple deviations from how the TPN compounders were placed and used inside of (not necessarily) a cleanroom layout. At a place, we visited we noticed the sink being placed right on the side of the open hood. But risks of particulate contamination don’t depend solely on how the room has been set up.
Risks associated with TPN compounding:
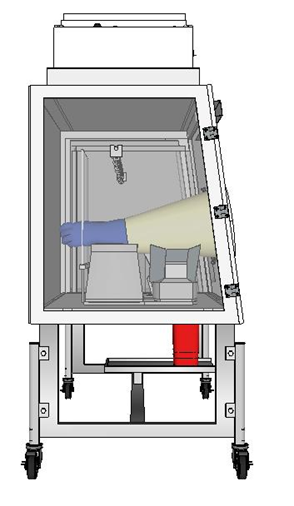
There are many aseptic risk factors when operating an Automated Pumping TPN Compounder in an open hood. For instance, we discovered that the TPN compounding machines are simply too big to be sitting inside of a LAWS. In a Laminar Air-Flow Work Station, you have the sterile air coming from the back, and the compounding machine itself is blocking the First Air, thus making what is supposed to be Uni-Directional Air flow – Turbulent. Another thing related to the size of the machinery and Its placement in an open hood is that the turbulent air disturbance causes drag and particulate accumulation on the backside equipment even with daily wipes and proper cleaning techniques.
The size of the TPN compounding machinery means that the compounder sits way past the safe 6-inch line to where First Air is effective. Practically you have components of the TPN machinery that is sticking outside of the LAWS. Pharmacy technicians are forced to use this equipment incorrectly just because it won’t fit inside a standard Open Hood properly.
Other serious risk factors are the huge potential for direct touch contamination. You have the operator place the IV bag and handle the injection ports while setting up the bag for filling. Then the technician was handling the touch commands giving the compounder instructions on what solution it needs to mix, and we previously stated that the touch screen is sitting way outside the Hood. We were looking for any protocols that instructed technicians on changing sterile gloves every time they’ll be using the touch commands before handling the injectable material.
Another design and use flaws when operating a TPN compounder in an open hood is the Barcode scanner which is rested inside of the hood, but can be moved around and is meant to be used on the inside and outside of the hood. We simply witnessed air from outside of the open hood swirling around that barcode scanner and getting all over the barcode scanner, and after using that tool, the technician places it on the prebuilt rest which is on the inside of the hood, which directly introduces particles to the inside of the hood.
The Massachusetts State Board of Pharmacy accompanied by Investigating entourage by the FDA performed a couple of observations of facilities that were preparing Total Parenteral Nutrition products using various TPN compounders. Just recently the FDA noted another flaw in operation that is applicable to nearly all TPN compounders and that can directly be linked to how the machinery sits in an open hood. In the past FDA inspectors reported that the TPN compounder itself were just too big to fit inside of the regular 41” laminar flow hood that it’s disrupting the laminar airflow inside the hood. At another point, at another facility, the FL State BOP inspector noticed the possibility for touch contamination during normal operations with the TPNs.
In a separate observation, the FDA reported inadequate HEPA filter coverage and airflow over the area to which sterile product was exposed. The inspectors noted that the ISO 5 Laminar Flow Hoods weren’t able to maintain unidirectional airflow within the hood to ensure that the sterility of the product is maintained. This was due to the fact that the TPN compounder unit with numerous accessories that comprise approximately 60% of the hood’s capacity.
The most recently released report of a site observation by the FDA note the following issues with the TPN machinery and how they are typically placed and used in the open Hood, which is a routine practice used for TPN processing operations. The setup of the machinery with huge proportions on its own, which contains a TPN unit with numerous accessories which comprise a huge portion of the hood’s capacity and without adequately documenting the airflow smoke pattern it cannot be determined that this setup would have an effect on the unidirectional airflow within the hood to ensure that the sterility of the product is maintained.
The size of the equipment and the limited access to the very last component and accessory inside of the hood also lead to the operator Pharmacy Technician that is sterile processing TPN products inconsistently puts the top of his head (exposing the skin from his neck and face) into the hood along with parts of the shoulders, arms and hands. Another aspect is that while processing TPN products within the open Hood his non-sterile sleeves came in contact with the aseptic connection for the filler and contaminating the sterility of the hood.
At many other oversights, the observers reported that many operators were handling the filled product inside of IV bags with bare hands and while the hood HVAC systems were switched off. That way the operator contaminated the aseptic environment releasing particles inside of the LAWH. They were also exposing their skin to direct risk of coming in contact with liquids that might leak off the bag during the filling process, which is an issue that is common for all automated TPN fillers when the pump needles are clogged with particles from liquids that are constantly passing through them.
It’s interesting how the 0TPN machine manufacturers do not specify where and how this equipment gets placed right now, and facilities are forced to consult with their advisors on how to properly use this machinery. There are more than Ten Thousand machines from various manufacturers that are installed improperly across the States.
Aseptic Enclosures has developed a custom enclosure, part of the newly introduced XLTC series that is designed specially to eliminate all of the previously specified aseptic risks. Our product houses the TPN inside of a positive pressure isolator and a custom made enclosure with a pass-through chamber that ensures that the equipment does not affect the airflow inside the engineering controls chamber.

Our XLTC TPN Enclosure eliminates all risks that are related to its position in the open hood as well as improving the overall usability of the machine. The Aseptic Enclosures TPN Compounding Isolator completely eliminates all risks from particulate air and touch contamination of the system, and strongly improves personnel safety by placing a barrier between the pharmacy technicians and the compounding process that is carried out by the TPN compounder. By having the machinery sitting in an isolator we are reducing the risks related to aseptic processing, eliminating the sources for touch contamination and improve the Uni-Directional First Air.

The Aseptic Enclosures XLTC TPN Compounding Isolator is a serious aseptic improvement to your parenteral nutrition preparations. It should be considered as a necessity in all future installs of such a compounding equipment and made a mandatory upgrade for machines currently in operation.
References:
WARNING LETTER 16-PHI-11 dated August 19, 2016 https://www.fda.gov/ICECI/EnforcementActions/WarningLetters/2016/ucm518429.htm
WARNING LETTER 2016-DAL-WL-19 dated April 25, 2016 issued to Lincare Holdings, Inc. https://www.fda.gov/iceci/enforcementactions/warningletters/2016/ucm500608.htm
WARNING LETTER 3009590582 issued to Central Admixture Pharmacy Services, Inc. https://www.fda.gov/downloads/aboutfda/centersoffices/officeofglobalregulatoryoperationsandpolicy/ora/oraelectronicreadingroom/ucm523457.pdf
January 28, 2016: Federal Criminal Charges Filed Against Two Pharmacists for Adulteration of Drugs in Connection with Alabama-Based Compounding Pharmacy https://www.fda.gov/iceci/criminalinvestigations/ucm484850.htm
WARNING LETTER 3004407863 issued to Central Admixture Pharmacy Services, Inc. https://www.fda.gov/downloads/aboutfda/centersoffices/officeofglobalregulatoryoperationsandpolicy/ora/oraelectronicreadingroom/ucm401435.pdf
New England Life Care, Inc., Woburn, MA. 483 Issued 02/08/2018https://www.fda.gov/downloads/AboutFDA/CentersOffices/OfficeofGlobalRegulatoryOperationsandPolicy/ORA/ORAElectronicReadingRoom/UCM599182.pdf


Hi I am Dr. Enas from elements pharmacy Muscat , Sultanate Oman
I am intersted in this kind of TPN Machine I would like to have a qutation please
please send me to my email: enas.hosny@tmdone.com or contact me through
0096894718497